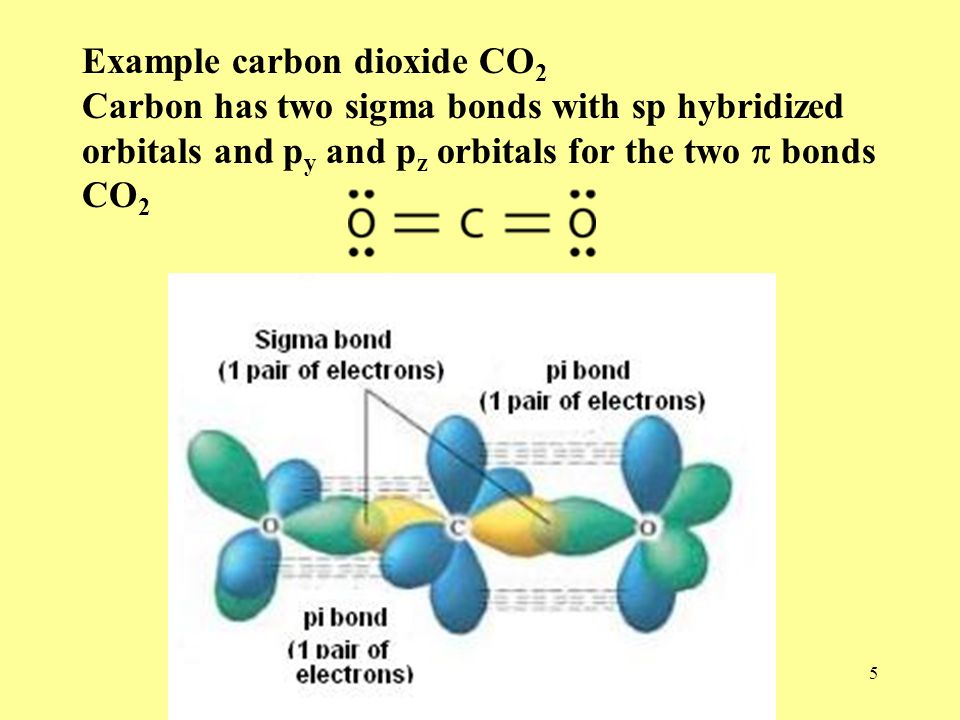
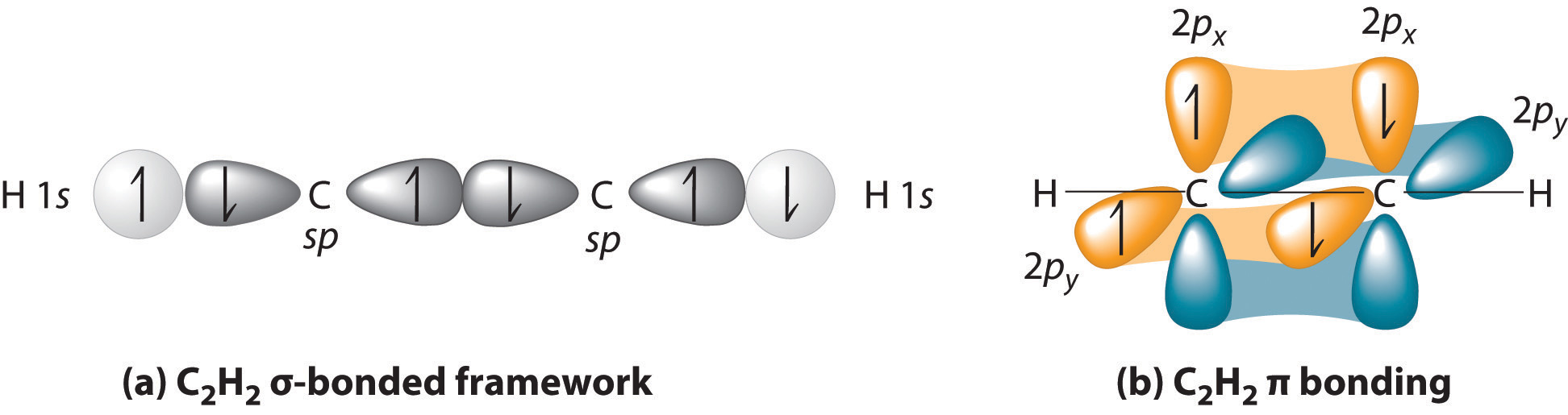
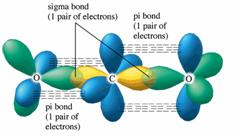
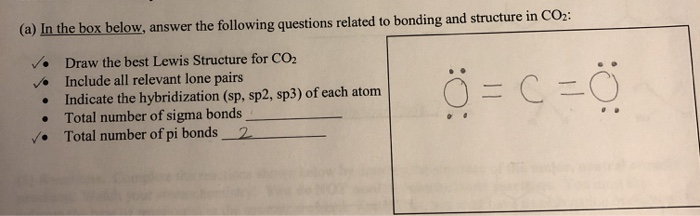
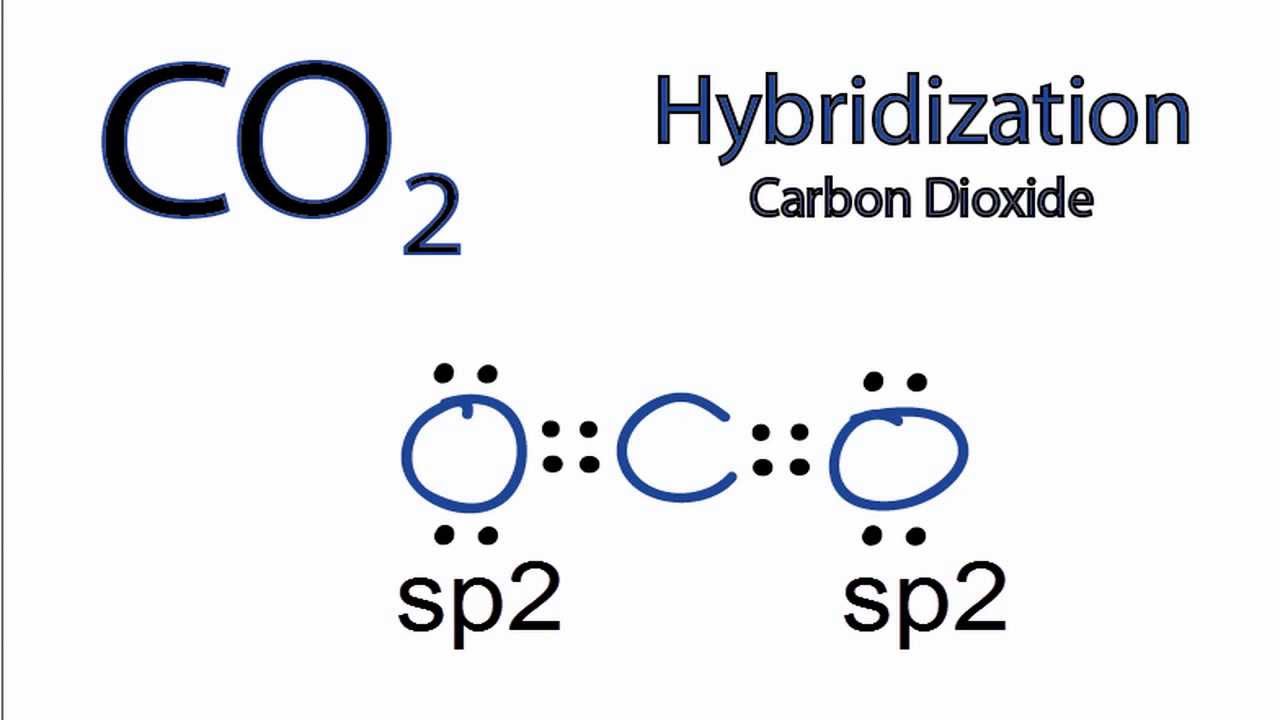
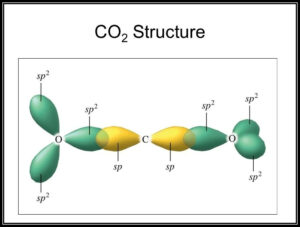
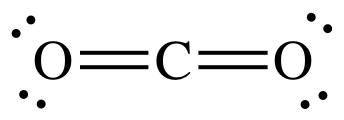What atomic or hybrid orbitals make up the pi bond between C and O in carbon dioxide, CO_2 ? orbital on C + orbital on O How many o bonds does C
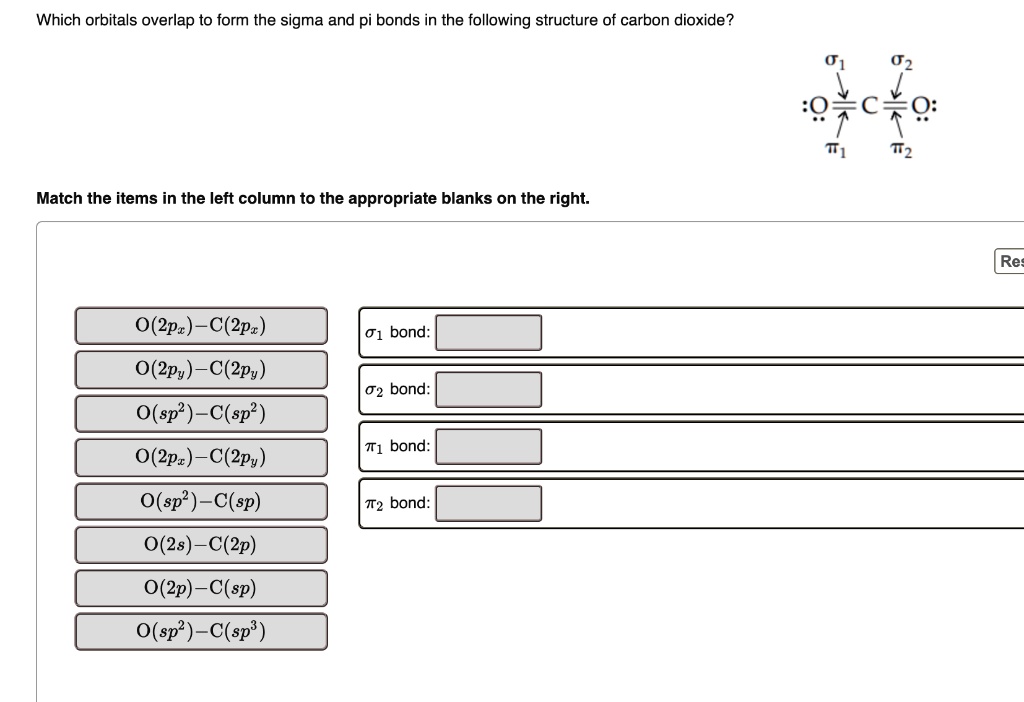
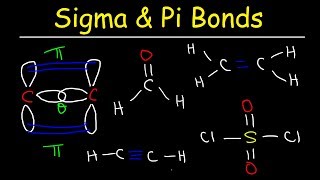
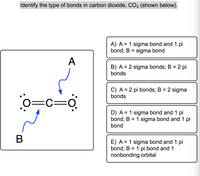
SOLVED: Which orbitals overlap to form the sigma and pi bonds in the following structure of carbon dioxide? 01 02 :0 C Q: T1 T2 Match the items in the left column
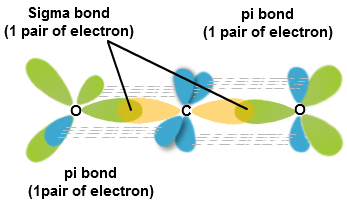
✓ Solved: Describe the bonding on the carbon atom in carbon dioxide, CO2, using valence bond theory.

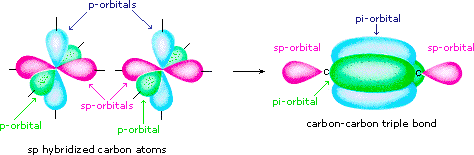
Allene is structurally related to carbon dioxide, CO2. Draw a picture showing the orbitals involved in the sigma and pi bonds of CO2, and identify the likely hybridization of carbon. | Homework.Study.com

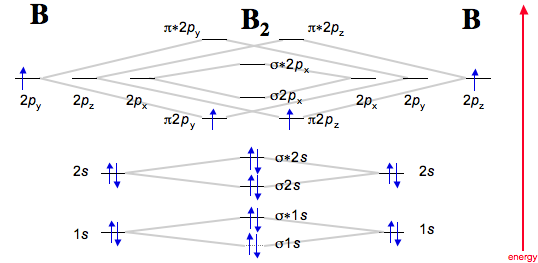
Molecular Orbital Diagram of Polyatomic CO2 Molecules - Chemical Bonding & Molecular Structures - YouTube